By Jim Riggs1
In the first article of this series, Jim discussed control relevant issues associated with distillation columns. The second article presented the major disturbances affecting composition control and the importance of properly functioning regulatory controls.
In this article, Jim discusses the use of product composition measurements in distillation column control. He also explores single composition strategies where either the top or bottom product composition is controlled while the other is allowed to float.
Product Composition Measurements
Product impurity levels (measured either on-line or in the laboratory) are used by feedback controllers to adjust column operation to meet product specifications. In addition, tray temperatures are used to infer product compositions, where feasible.
On-line analyzers
There is a range of on-line analyzers commonly used in the chemical process industries (e.g., gas chromatographs). A key issue with analyzers is their associated analyzer delay.
For columns that have slow-responding composition dynamics, analyzer delay is usually less of an issue. For a propylene/propane splitter (i.e., a high reflux ratio column), the composition dynamics for the primary product have a time constant of about 2 hours. As the cycle time for the analyzer increases from 5 minutes to 10 minutes, it does not significantly affect the feedback control performance.
For an ethane/propane splitter (i.e., a low reflux ratio column), even a five-minute analyzer delay seriously undermines composition control performance because the time constant for this process is less than five minutes. Fortunately, most fast-acting columns have a significant temperature drop across them so that product composition can be effectively inferred from tray temperature measurements.
For columns that have a low relative volatility (i.e., less than 1.4), inferential temperature control is not feasible and feedback based on an on-line analyzer is required, but these columns generally have slow composition dynamics compared to the analyzer delay. For columns with a high relative volatility (i.e., greater than 2.0), inferential temperature control is usually effective.
Inferential temperature control
Inferential temperature control is an effective means of maintaining composition control for columns from a control and economic standpoint. That is, it greatly reduces the analyzer dead time for feedback control and is much less expensive to install and maintain than an on-line composition analyzer. Because of their superior repeatability, RTDs (resistance temperature detectors) and thermistors are usually used for inferential temperature applications.
For multicomponent separations, tray temperatures do not uniquely determine the product composition. As a result, for these cases it is essential that an on-line analyzer or, at least, periodic laboratory analysis be used to adjust the tray temperature set point to the proper level.
If feedback based on laboratory analysis is not used, offset between the desired product composition and the actual product composition can result. Column pressure also significantly affects tray temperature measurements. For most systems, a simple linear correction compensates for variations in column pressure:
where Tpc is the pressure-compensated temperature that should be used for feedback control, Tmeas is the measured tray temperature, Kpr is pressure correction factor, P is the operating pressure and P0 is the reference pressure.
Kpr can be estimated by applying a steady-state column simulator for two different pressures within the normal operating range and using the following equation:
where Ti is the value of the tray temperature for tray i predicted by the column simulator.
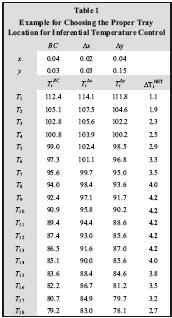
A steady-state column model can also be used to determine the best tray locations for inferential control by finding the trays whose temperatures show the strongest correlation with product composition. The following procedure is recommended:
Table 1 (click for a large view) shows an example of this approach used to locate the best tray temperature for inferential temperature control in the stripping section of a depropanizer.
Note that a temperature measurement anywhere between tray 7 and tray 16 should work well for this application for the assumed feed composition.
For certain columns, the bulk of the temperature change occurs in a few trays, resulting in a very steep temperature profile. If a single tray temperature is used to infer the product composition in such a case, feed composition changes can move the location of the steep temperature change away from the tray selected for control, leading to a situation in which the chosen tray temperature is insensitive to changes in product impurity levels.
This problem can be handled by controlling the average of several tray temperatures that bracket the area where the steep temperature changes occur for feed composition changes. In this manner, when feed changes cause the temperature profile in the column to move, at least one of the tray temperatures used in the average of the temperatures is located on the steep temperature front. The average of the tray temperatures should still be sensitive to product impurity changes over the full range of feed composition changes.
Single Composition Control
Here the composition of one product is controlled while the composition of the other product is allowed to float. In the chemical industry over 90% of the columns are operated under single composition control compared to dual composition control, which controls both the overhead and the bottoms product compositions.
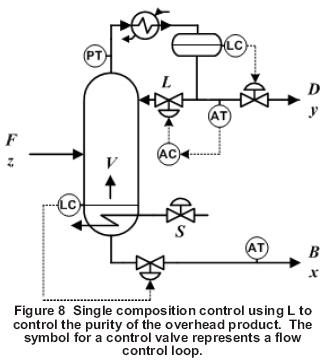
Figure 8 shows single composition control using the reflux to control the purity of the overhead product while maintaining a fixed reboiler duty. The bottom composition is not controlled directly and can vary significantly as the feed composition changes.
The control performance of the overhead product is generally best when reflux, L, rather than either the distillate flow rate, D, or the reflux ratio, L/D is the MV. L is the fastest-acting MV for the overhead and the least sensitive to feed composition changes. Because the reboiler duty is fixed, coupling is not an issue
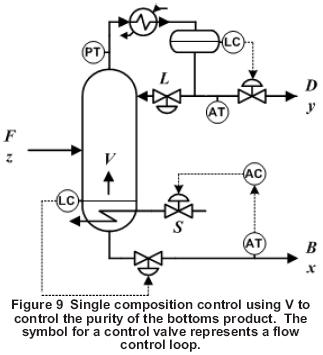
When the bottom product is controlled by single composition control, the control configuration shown in Figure 9 is recommended. Because the boilup rate, V, is faster acting and less sensitive to disturbances than either the bottoms product rate, B, or the boilup ratio, V/B, V is used to control the bottom product composition with the reflux rate fixed, which allows the overhead composition to float.
Single composition control is much easier to implement, tune and maintain than dual composition control. The choice between single and dual composition control is based on the tradeoff between the additional cost associated with dual composition control (analyzer costs, increased controller maintenance, etc.) and the economic benefit of dual composition control (increased product recovery and reduced utility costs).
While single composition control is in widespread use in the chemical industry, dual composition control is generally preferred for refinery columns and columns that produce high-volume chemical intermediates because the energy usage for these columns is much larger.
More on Distillation Control
The final article in this four part series on distillation control will discuss advanced issues including dual composition control and constraint control.
_______
1. This material is reprinted from Chemical Process Control, 2nd Ed. with the permission of the publisher: Ferret Publishing (806 747 3872).
About the Author
Jim Riggs is a professor of chemical engineering at Texas Tech University, where he has been since 1983. He has served as an industrial consultant and presented a number of industrial short courses on various topics relating to process control. He is the author of several popular chemical engineering textbooks and co-founded the Texas Tech Process Control Consortium in 1992.
Jim Riggs
Department of Chemical Engineering
Texas Tech University
Lubbock, Texas 79410
Email: jim.riggs@ttu.edu